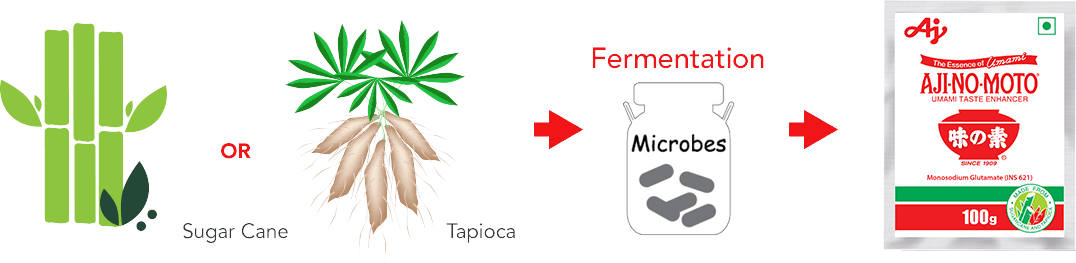
- Umami seasoning AJI-NO-MOTO® is a brand name of Umami Seasoning product (Monosodium glutamate, MSG), which is made from the finest quality crops like sugarcane and tapioca through natural fermentation process.
- Monosodium glutamate (MSG) is a flavour enhancer which has been used effectively for over a century to bring out the best flavour of food.

Safety of Umami seasoning
AJI-NO-MOTO®
What Is Umami seasoning AJI-NO-MOTO®?
Raw Material Used To Produce Umami seasoning AJI-NO-MOTO®:
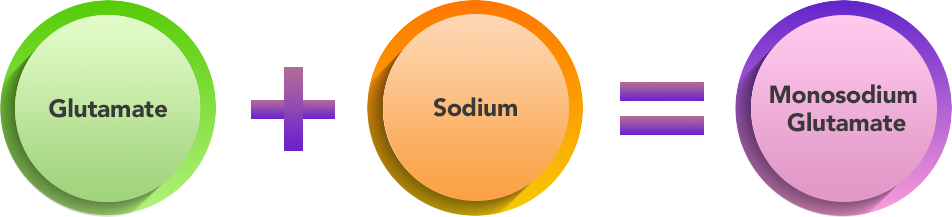
- MSG is the combination of glutamate and sodium.
Components Of MSG :
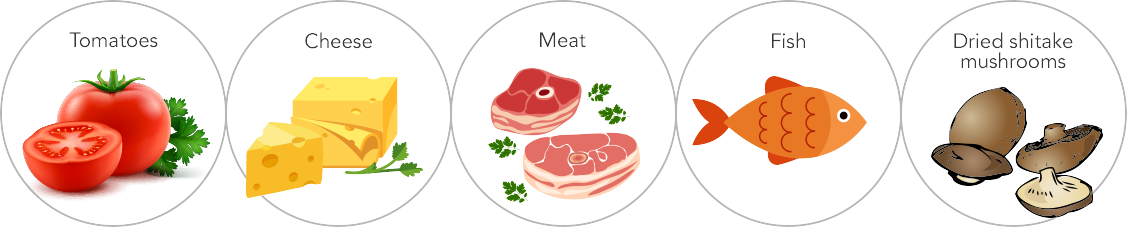
- Glutamate is one of the amino acids which is found abundantly in most natural foods such as tomato, onion, cheese and milk etc.
- Sodium is one of the essential minerals which is needed for the daily functioning of the human body.
Natural Sources Of Glutamate :
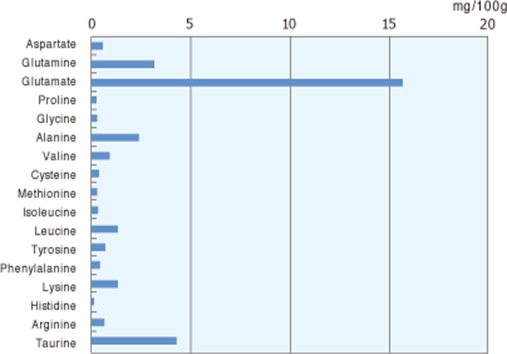
- Even mother breast milk also contains glutamate content in it.
Free Amino Acids In Human Breast Milk :
TRUTH ABOUT UMAMI SEASONING AJI-NO-MOTO®
Safety Assurance
Since 1909, Umami seasoning AJI-NO-MOTO® seasoning has been safely used throughout the world and it is also one of the most extensively researched food ingredients with more than hundreds of Scientifics studies affirmed the safety of MSG consumption. Below are the lists of organizations that have declared the safety of MSG consumption: such as tomatoes and mushrooms, have high levels of naturally occurring free glutamate.

FSSAI
Subjected to Good manufacturing practices (GMP) no defined level prescribed in the FSSAI act, FSSAI Clause No: 3.1.11 Sub Class No: (XXIII): Use OF flavor Enhancer (MSG) in following food items is allowed – Noodles seasoning and pasta, meat tenderizer, onion salt, garlic salt, oriental seasoning mix etc. In Table 10 referring to FSSAI in the Seasoning Mix, culinary powder category dosage of MSG is given as GMP.

US FDA
In 1995, the Federation of American Societies for Experimental Biology (FASEB) reaffirmed the safety of MSG for the general population. In its review, commissioned by the FDA, FASEB found no evidence linking MSG to any serious or long-term health effects, which led the FDA to again reaffirm that MSG is a safe food ingredient at normally consumed levels.

FS of Australia New Zealand
Conclusion reached by Foods Standards Australia New Zealand: MSG is safe In 2002, Food Standards Australia New Zealand conducted a review of the safety of MSG. The Chief Scientist concluded that several recent reviews of the scientific evidence have confirmed that MSG is safe for the general public at the levels of use typically found in food.

European Countries
European Communities: ADI not specified In 1991, the European Commission’s Scientific Committee for Food (SCF) also affirmed MSG’s safety. Having reviewed the most advanced and up-to-date research on glutamate, the SCF published a report in 1991 which designated an ‘ADI not specified’ for MSG.

JECFA
Conclusion made by JECFA: ADI not specified JECFA* evaluated the safety of glutamate in 1970, 1973 and 1987.After three safety evaluations, JECFA placed MSG in the safest category, “Acceptable Daily Intake (ADI) not specified” * JECFA is a prestigious scientific advisory body to the World Health Organization (WHO) and the Food and Agricultural Organization (FAO) of the United Nations.

American Academy of Allergy, Asthma & Immunology (AAAAI)
A professional organization with more than 6,700 members in the United States, Canada and 72 other countries with a special interest in the research and treatment of allergic and immunologic diseases (For more info:- http://www.aaaai.org/about-aaaai)
AAAAI has concluded that –“There were no reactors in MSG found to be associated with the provocation of Asthma attack”
(Read article about Food Allergy: A Practice Parameter)
Link:- http://www.aaaai.org/Aaaai/media/MediaLibrary/PDF%20Documents/Practice%20and%20Parameters/food-allergy-2006.pdf





